How to Export Pharmaceuticals from India — Complete Guide
India is the pharmacy of the world. In 2024-25, India's pharmaceutical exports reached $22 billion, a 32% surge from $16.7 billion just two years earlier. Indian pharma companies supply 40% of the generic medicines consumed in the United States, 25% of all medicines in the UK, and 50% or more of vaccines globally. For MSME pharmaceutical manufacturers, the export opportunity is enormous — but so is the regulatory complexity.
Pharmaceutical exports are the most heavily regulated category in international trade. Every destination country has its own drug regulatory authority, its own approval process, and its own standards for manufacturing quality. A single compliance failure can result in import bans, FDA warning letters, or criminal prosecution. This guide covers the regulatory pathway, key markets, quality requirements, and practical steps for Indian pharma MSMEs looking to export.
India's Pharmaceutical Export Landscape
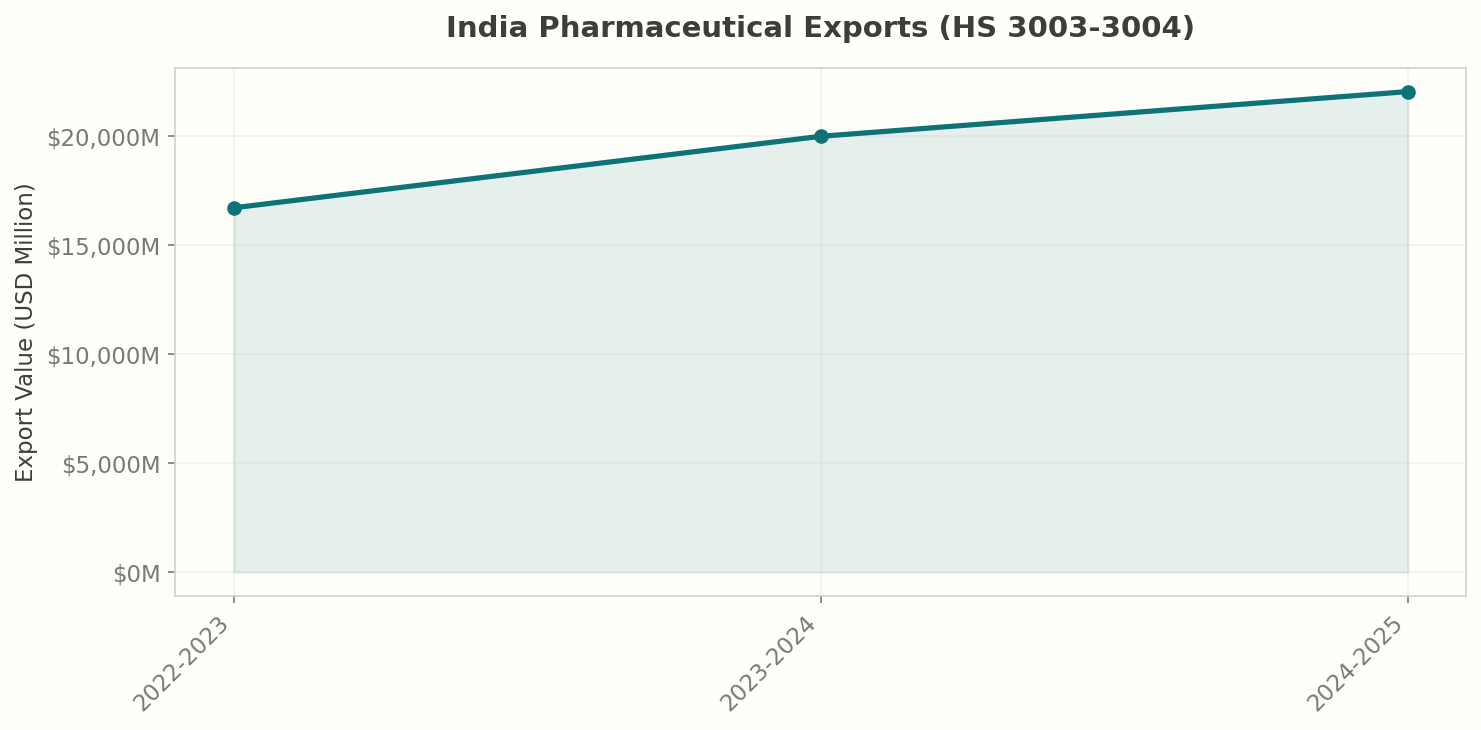
India exported $22 billion in pharmaceutical products (HS 3003-3004) in 2024-25, making it the sector's strongest year on record. The growth trajectory has been remarkable:
| Year | Pharma Exports (USD Billion) | Growth |
|---|---|---|
| 2022-23 | $16.7 | — |
| 2023-24 | $20.0 | +19.6% |
| 2024-25 | $22.0 | +10.3% |
India accounts for approximately 20% of global generic drug production by volume. The country has over 3,000 pharma companies and over 10,500 manufacturing units. More importantly, Indian facilities hold the largest number of US FDA-approved plants outside the USA — over 680 facilities.
Where Indian Pharmaceuticals Are in Demand
India is known as the "pharmacy of the world," and Indian pharmaceutical exports reach over 200 countries. The USA is overwhelmingly the largest market, driven by generic drug demand — India supplies approximately 40% of all generic prescriptions dispensed in America. African markets collectively represent a large and growing destination for affordable Indian medicines. European markets, Russia, and South Asia are also significant. India's pharmaceutical export strength lies in generics, APIs (active pharmaceutical ingredients), and increasingly in biosimilars and complex generics.
Want the full country-by-country breakdown? See exact export values, growth rates, tariff rates, and market attractiveness scores for every destination in our detailed data pages. View Pharmaceuticals export data by country →
Regulatory Framework — Who Approves What
In India — CDSCO
The Central Drugs Standard Control Organisation (CDSCO) is India's national drug regulatory authority. Before exporting, your pharmaceutical product needs:
Drug Manufacturing Licence: Issued by the State Drug Controller (for most dosage forms) or CDSCO Central (for blood products, vaccines, biologicals). Your manufacturing facility must meet Schedule M of the Drugs and Cosmetics Act.
Export Registration Certificate: For formulations exported to countries that require it, CDSCO issues a Certificate of Pharmaceutical Product (CoPP) in the WHO format. This certificate confirms that your product is approved for sale in India and that your facility meets GMP requirements.
No Objection Certificate (NOC) for Export: Required for certain controlled substances and bulk drugs under export restriction.
For the USA — FDA
The US FDA is the gold standard in pharmaceutical regulation:
ANDA (Abbreviated New Drug Application): To sell a generic drug in the USA, you must file an ANDA with the FDA. This requires demonstrating bioequivalence with the Reference Listed Drug (RLD). Timeline: 12-18 months for approval, but backlog can extend to 2-3 years.
Facility Registration: Your manufacturing facility must be registered with the FDA and assigned a Facility Establishment Identifier (FEI). Registration is annual and done through the FDA's Unified Registration and Listing System.
cGMP Compliance: FDA inspects manufacturing facilities — both pre-approval (before ANDA approval) and routine inspections. The inspection checks compliance with 21 CFR Parts 210/211 (cGMP for finished pharmaceuticals). Non-compliance results in Form 483 observations, Warning Letters, and potentially Import Alerts (ban on import).
Drug Listing: Every drug product exported to the USA must be listed in the FDA's Drug Listing System with its National Drug Code (NDC).
For the EU — EMA/National Authorities
Marketing Authorisation: Through either the centralised procedure (EMA) or national procedures (individual country agencies). The decentralised procedure allows approval in multiple EU countries simultaneously.
EU GMP Certification: Manufacturing facilities must have a valid EU GMP certificate issued after inspection by an EU authority or their delegate. Indian facilities exporting to the EU are inspected by EU regulatory agencies.
Written Confirmation (Annex 16): For APIs (Active Pharmaceutical Ingredients) exported to the EU, a Written Confirmation from CDSCO is required, confirming that the facility meets standards equivalent to EU GMP.
For Regulated Markets — WHO GMP
WHO Prequalification: For supplying to UN agencies (UNICEF, WHO, Global Fund), your product needs WHO Prequalification. This involves dossier review and facility inspection against WHO GMP standards. Indian companies hold the highest number of WHO Prequalified products globally.
WHO GMP Certificate: Even for non-prequalified exports, many countries in Africa, Asia, and Latin America require a WHO-format GMP certificate from CDSCO as a baseline quality assurance.
HS Codes for Pharmaceutical Exports
| HS Code | Description |
|---|---|
| 3003 | Medicaments (not in dosage form, not for retail sale) — bulk drugs and intermediates |
| 3004 | Medicaments in measured doses or packed for retail sale — finished formulations |
| 300410 | Containing penicillins or streptomycins |
| 300420 | Containing antibiotics |
| 300431 | Containing insulin |
| 300432 | Containing adrenal cortex hormones |
| 300450 | Containing vitamins |
| 300490 | Other medicaments (catch-all for most generics) |
Most Indian pharma exports fall under 300490 — the "other" category that covers the bulk of generic formulations. Use the HS Code Finder for precise classification of your product.
Quality Systems and Manufacturing Standards
Schedule M (India GMP)
The revised Schedule M (effective June 2024) significantly upgraded India's GMP standards to align more closely with WHO GMP. Key requirements:
- Quality Management System with a dedicated Quality Assurance unit
- Validation of manufacturing processes and analytical methods
- Stability studies (accelerated and long-term)
- Clean room classification for sterile products
- Water purification system qualification
- Equipment calibration and preventive maintenance
ICH Guidelines
The International Council for Harmonisation (ICH) guidelines are increasingly adopted by Indian regulators. Key guidelines relevant to exporters:
- ICH Q7 — GMP for APIs
- ICH Q1 — Stability testing (Q1A for new drug substances, Q1B for photostability, Q1E for evaluation of stability data)
- ICH Q2 — Analytical method validation
- ICH Q6A — Specifications for new drug substances
- ICH Q9 — Quality risk management
- ICH Q10 — Pharmaceutical quality system
Testing Infrastructure and Costs
Quality control testing is a significant ongoing cost for pharma exporters. Key NABL-accredited laboratories for pharmaceutical testing include Central Drug Laboratory (CDL, Kolkata), Indian Pharmacopoeia Commission (IPC, Ghaziabad), and private labs such as SGS India, Eurofins Pharma, Vimta Labs (Hyderabad), and Lambda Therapeutic Research (Ahmedabad). Bioequivalence studies for an ANDA filing cost Rs 30-80 lakh per study depending on the drug and study design. Stability testing (6-month accelerated + 12-month long-term) costs Rs 2-5 lakh per product. Routine batch release testing (assay, dissolution, content uniformity, related substances, microbiological limits) costs Rs 15,000-50,000 per batch. For an MSME planning to file 2-3 ANDAs per year, budget Rs 1-2 crore annually for quality control and regulatory testing alone.
Data Integrity
Since 2015, data integrity has been the most common reason for FDA Warning Letters to Indian pharma companies. FDA expects:
- Complete, original, accurate, and attributable data (ALCOA+ principles)
- Audit trails for all electronic records
- No manipulation, deletion, or back-dating of manufacturing or testing data
Export Documentation for Pharmaceuticals
Standard export documents plus pharma-specific requirements:
- Commercial Invoice
- Packing List
- Shipping Bill
- Bill of Lading / Airway Bill
- Certificate of Origin
- Certificate of Pharmaceutical Product (CoPP) — from CDSCO
- WHO GMP Certificate — from CDSCO
- Free Sale Certificate — confirming the product is freely sold in India
- Certificate of Analysis (CoA) — batch-specific quality analysis report
- Stability Data — if required by the importing country
- Product Dossier — regulatory submission to the importing country's drug authority
- Insurance Certificate (if CIF)
Cold chain documentation: For temperature-sensitive products (vaccines, biologicals, insulin), include temperature monitoring records (data loggers) and cold chain compliance certificates.
Logistics for Pharmaceutical Exports
Temperature Control
Pharmaceuticals require strict temperature management:
- Ambient (15-25°C): Most solid oral dosage forms (tablets, capsules)
- Cold chain (2-8°C): Vaccines, biologicals, insulin, certain injectables
- Frozen (-20°C): Some biologicals and diagnostic reagents
For cold chain products, use GDP-certified logistics providers (Good Distribution Practice). Major options: DHL Life Sciences, FedEx Healthcare, UPS Temperature True.
Packaging and Labelling
- Primary packaging: Blister packs (alu-alu or alu-PVC), bottles (HDPE, glass)
- Secondary packaging: Cartons with product information leaflet
- Tertiary packaging: Corrugated shippers, palletised for container loading
- Serialisation: Many countries (USA, EU, Saudi Arabia) require serialisation (unique identification codes on each unit) for drug traceability
Region-specific packaging requirements:
- USA: FDA requires tamper-evident packaging for OTC drugs. NDC number, drug name, strength, dosage form, manufacturer name and address, lot number, expiry date, and adequate directions for use must appear on the label. Child-resistant packaging is required for most prescription drugs under the Poison Prevention Packaging Act.
- EU: Falsified Medicines Directive (FMD) mandates unique identifiers (2D data matrix codes) and tamper-evident features on prescription medicines. Patient information leaflets must be in the official language(s) of the destination country. Braille must appear on outer packaging.
- Middle East/Africa: Arabic labelling is mandatory in GCC countries. Many African nations accept English-language labels but require the WHO International Nonproprietary Name (INN) on the packaging. Saudi Arabia's SFDA requires specific serialisation under the RSD (Rassd) track-and-trace system.
Shipping
- Air freight: Preferred for high-value, low-volume shipments and all cold chain products. Mumbai and Hyderabad airports have dedicated pharma handling zones.
- Sea freight: For large-volume, ambient-temperature products. 20-ft container holds approximately 22-24 tonnes of pharmaceutical products.
Government Support
Pharma export promotion: Pharmaceuticals Export Promotion Council (Pharmexcil) is the designated export promotion council. Membership provides access to trade delegations, buyer-seller meets, and market intelligence. Pharmexcil organises the annual International Pharmaceutical Exhibition (IPHEX) and sponsors Indian pavilions at CPhI Worldwide (Frankfurt/Milan), Arab Health (Dubai), and Africa Health (Johannesburg).
PLI Scheme for Pharmaceuticals: Production-Linked Incentive scheme covering bulk drugs (APIs) and medical devices. Incentives of 5-20% of incremental sales for specified products. PLI-I covers 53 critical bulk drugs/KSMs (Key Starting Materials) with 20% incentive on incremental sales. PLI-II covers complex generics, biologics, and medical devices with 5-10% incentive.
WHO GMP/COPP facilitation: CDSCO has streamlined the process for WHO GMP and CoPP issuance, with online application through the SUGAM portal. Typical processing time is 3-6 weeks for CoPP and 2-4 months for WHO GMP inspection.
RoDTEP: Pharmaceutical products under HS 3004 attract RoDTEP rates of 0.5-2% of FOB value. Check the DGFT RoDTEP schedule for your specific 8-digit tariff code.
Duty Drawback: Rates of 1.5-3% apply to finished formulation exports, refunding customs duties paid on imported excipients and packaging materials.
Export Credit: Pharma exports qualify for pre-shipment and post-shipment finance at concessional interest rates under RBI's priority sector lending guidelines. ECGC provides export credit insurance covering commercial risks (buyer default) and political risks (war, currency inconvertibility). EXIM Bank offers lines of credit for pharma exports to African and Asian countries, which can facilitate large government procurement contracts.
Finding Buyers and Building Market Access
International trade shows: CPhI Worldwide (the largest pharma ingredients show, held annually in Europe), Arab Health (Dubai, January), Medica (Dusseldorf, November), Africa Health (Johannesburg), and CPHI India (Greater Noida) are the key platforms for meeting international buyers, distributors, and tender agents.
B2B portals and tender platforms: Register on Pharmexcil's Trade Directory, PharmaCompass, and global tender platforms such as UNGM (United Nations Global Marketplace), UNICEF Supply Division, and Global Fund procurement portals. Many African government tenders are listed on dgMarket and national procurement agency websites.
Embassy commercial sections: Indian embassies in target countries maintain commercial wings that can introduce you to local distributors and regulatory consultants. Request a meeting through the Trade Promotion Division of the Ministry of External Affairs or directly contact the embassy's commercial attache.
Registration agents: In most regulated markets, you need an in-country registration agent or marketing authorisation holder. For African markets, companies like PharmAccess, Ubipharm, and local pharmacy distributors serve as registration partners. For the EU, you need a Qualified Person (QP) for batch release and an EU-based marketing authorisation holder.
Common Mistakes to Avoid
Underestimating FDA inspection preparation. An FDA inspection typically lasts 5-10 days. Companies that scramble to prepare only after inspection is announced invariably receive observations. Maintain inspection-readiness year-round.
Data integrity failures. This remains the biggest risk. If FDA finds evidence of data manipulation during an inspection, the consequences are severe — Import Alert, Warning Letter, and potential criminal referral.
Not maintaining post-market surveillance. Exporting a drug is not a one-time event. You must maintain adverse event reporting, stability monitoring, and annual product quality reviews for as long as the product is in the market.
Ignoring tender markets. Government tenders in Africa and Southeast Asia account for a large share of generic drug procurement. Many MSMEs focus only on the USA and ignore these volume opportunities. Register on UN agency procurement portals (UNICEF Supply Division, WHO) and national tender databases.
Shipping without adequate temperature monitoring. Temperature excursions during transit are a leading cause of pharmaceutical product degradation. Use calibrated data loggers in every shipment, even for ambient-temperature products. For cold chain products, ensure continuous temperature monitoring with real-time alerts. A single documented excursion above 25 degrees C for ambient products, or above 8 degrees C for cold chain products, can trigger rejection at the destination.
Not budgeting for regulatory timelines. Drug registration in a new country takes 6-24 months depending on the market. ANDA approval in the USA averages 18-24 months. WHO Prequalification takes 12-18 months. African regulatory approvals range from 3-12 months. Plan your market entry timeline and cash flow accordingly — revenue from a new market will not materialise for 1-2 years after you start the registration process.
Key Takeaways
- India's pharma exports hit $22 billion in 2024-25 — a 32% increase over two years
- FDA approval requires ANDA filing, facility registration, cGMP compliance, and regular inspections
- WHO GMP certificate from CDSCO is the baseline for most non-regulated markets
- Data integrity is the #1 risk factor for Indian pharma exporters — invest in ALCOA+ compliant systems
- Cold chain logistics are critical for vaccines and biologicals
Start Your Pharma Export Journey
- Verify your HS code with the HS Code Finder — most formulations fall under 300490
- Ensure your facility has WHO GMP certification from CDSCO — this is the minimum baseline for any export market
- Choose your target market — use the Market Finder to see demand by country
- Register with Pharmexcil for your RCMC and access to export promotion support
- Check country-specific regulatory requirements — guides for USA, UK, and other markets outline the drug registration pathway
- Invest in quality systems — GMP compliance, data integrity, and validation are not costs, they are investments that unlock market access
India's pharma sector has earned its reputation as the world's generic medicine supplier. With the right regulatory preparation and quality commitment, your facility can be part of this $22 billion success story.
Ready to start exporting?
Get a detailed market report with country-by-country analysis, pricing insights, and buyer contacts.
Get Market ReportXIMPEX Export Advisory
Need help implementing the strategies discussed in this guide? Our team of export specialists can provide personalized guidance for your business.