How to Export Surgical Instruments from India — Complete Guide
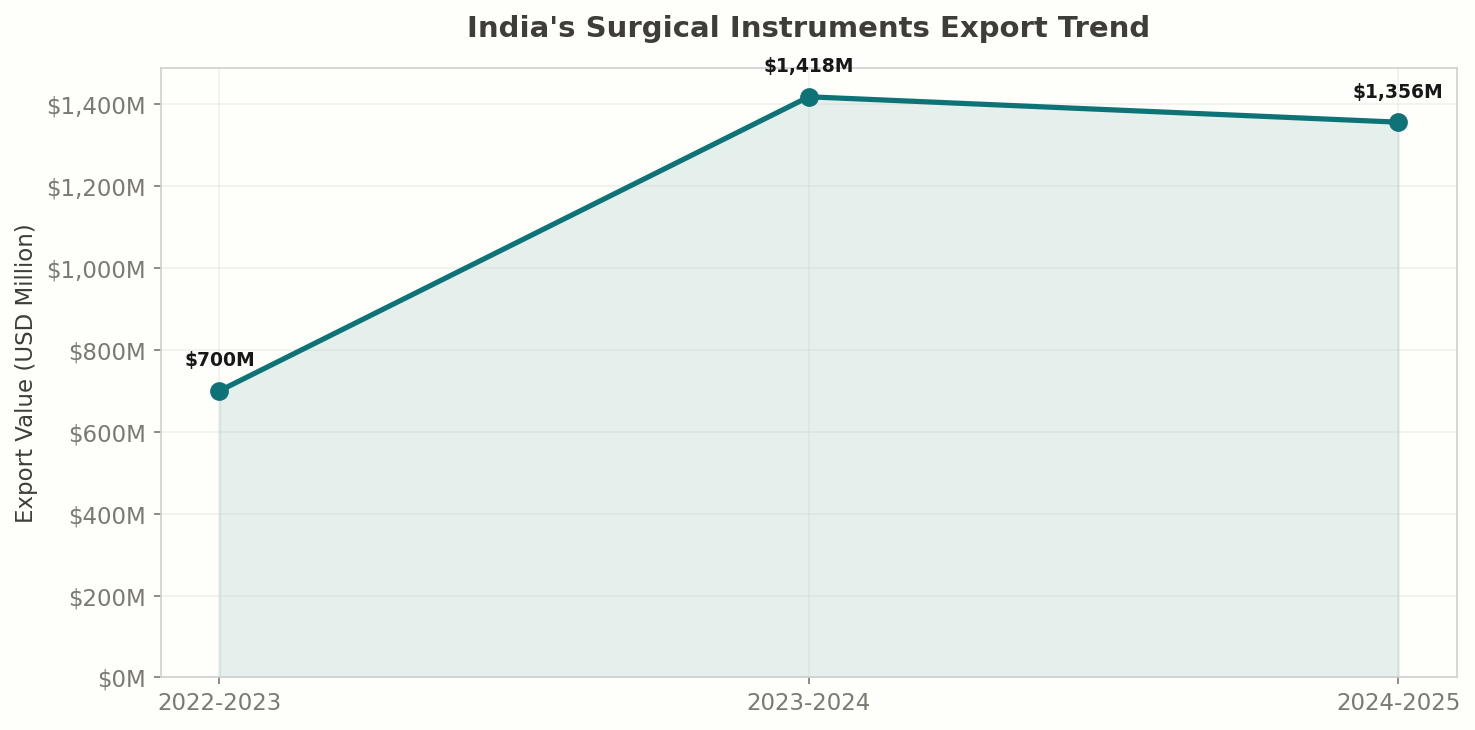
India's medical device and surgical instrument sector has undergone a remarkable transformation. In 2024-25, India exported $1,355.74 million (approximately $1.36 billion) in surgical and medical instruments under HS heading 9018, nearly doubling from $700 million in 2022-23. This 94% surge in two years reflects India's growing capability in medical device manufacturing, expanding FDA-approved facility base, and the global push to diversify medical supply chains beyond traditional sources.
For MSME manufacturers — whether you produce disposable syringes in Faridabad, orthopaedic implants in Pune, surgical sutures in Chennai, or diagnostic equipment in Bangalore — the export opportunity is enormous. Global healthcare spending exceeds $9 trillion, and the medical device market alone is worth $600 billion. India's advantages include competitive manufacturing costs (40-60% lower than developed markets), a large pool of biomedical engineers, growing R&D capability, and an expanding base of facilities certified to ISO 13485 and US FDA standards. However, this is the most regulated product category in international trade. A single quality failure can result in patient harm, product recalls, regulatory sanctions, and criminal liability. This guide covers the regulatory pathway, quality systems, market access strategies, and practical logistics for exporting surgical instruments from India.
India's Surgical Instrument Export Landscape
India exported $1,355.74 million in surgical and medical instruments (HS 9018) in 2024-25, making it one of the fastest-growing export categories in the country:
| HS Code | Category | 2024-25 Exports (USD Million) | Share |
|---|---|---|---|
| 901839 | Syringes, needles, catheters, cannulae | $512.7 | 37.8% |
| 901890 | Other instruments and appliances | $478.6 | 35.3% |
| 901813 | MRI, CT scanners (magnetic resonance/scintigraphic) | $154.8 | 11.4% |
| 901819 | Other electro-diagnostic apparatus | $68.8 | 5.1% |
| 901832 | Tubular metal needles, sutures | $50.2 | 3.7% |
| 901812 | ECG apparatus (electro-cardiographs) | $38.9 | 2.9% |
| 901850 | Ophthalmic instruments and appliances | $34.6 | 2.6% |
Syringes, needles, and catheters (HS 901839) dominate at $512.7 million, reflecting India's massive disposable medical device manufacturing capacity — the country produces over 4 billion syringes annually. "Other instruments and appliances" (HS 901890) at $478.6 million is a broad category covering surgical hand instruments, endoscopic accessories, implant delivery systems, and specialised surgical tools. High-value diagnostic equipment — MRI/CT scanners ($154.8M) and other electro-diagnostic devices ($68.8M) — represent India's growing capability in medical electronics.
Where Indian Surgical Instruments Are in Demand
The USA is the largest export market for Indian surgical and medical instruments, driven by demand for disposable medical devices, syringes, catheters, and diagnostic equipment. European markets — the Netherlands, Germany, and Belgium — are the second major cluster, reflecting India's growing role as a supplier to European healthcare systems. Emerging markets across Asia (China, Singapore), Latin America (Brazil), the Middle East (UAE, Turkey), and Russia represent a significant and growing share, driven by expanding healthcare infrastructure and India's cost-competitive manufacturing.
Want the full country-by-country breakdown? See exact export values, growth rates, tariff rates, and market attractiveness scores for every destination in our detailed data pages. View Surgical Instruments export data by country →
Product Categories
Disposable Medical Devices (HS 901839)
India is a global leader in disposable medical device manufacturing:
- Syringes — hypodermic, insulin, auto-disable (AD), prefilled
- Needles — hypodermic, spinal, biopsy, IV cannulae
- Catheters — urinary (Foley), central venous, angiography, guiding
- IV sets — infusion sets, blood transfusion sets, extension lines
- Blood collection systems — vacuum tubes, blood bags
Surgical Hand Instruments (HS 901890)
- General surgery — scissors, forceps, retractors, needle holders, scalpel handles
- Orthopaedic — bone saws, chisels, rongeurs, bone-holding forceps
- Laparoscopic — trocars, graspers, scissors, clip appliers
- Dental — extraction forceps, elevators, probes, scalers
- ENT — speculums, suction tips, curettes
Diagnostic Equipment (HS 901812, 901813, 901819)
- ECG machines — 3-lead, 12-lead, portable, telemetry
- Patient monitors — multi-parameter, pulse oximeters, capnographs
- MRI systems — permanent magnet, superconducting (emerging category)
- Ultrasound — portable, point-of-care, colour Doppler
Sutures and Needles (HS 901832)
- Absorbable sutures — polyglycolic acid (PGA), polyglactin (Vicryl equivalent)
- Non-absorbable sutures — silk, nylon, polypropylene, stainless steel
- Surgical needles — cutting, round body, taper point
- Staplers — skin staplers, surgical stapling devices
Ophthalmic Instruments (HS 901850)
- IOLs (intraocular lenses) — India is a major IOL manufacturer
- Surgical instruments — phaco handpieces, vitrectomy systems, microsurgical tools
- Diagnostic — tonometers, slit lamps, fundus cameras
HS Code Classification
| 6-Digit Code | Description | Key Products |
|---|---|---|
| 901811 | Electro-cardiographs | ECG machines, Holter monitors |
| 901812 | Ultrasonic scanning apparatus | Ultrasound systems, Doppler |
| 901813 | MRI, scintigraphic apparatus | MRI machines, CT scanners, PET |
| 901814 | Scintigraphic apparatus | Gamma cameras, SPECT |
| 901819 | Other electro-diagnostic apparatus | Patient monitors, EEG, EMG |
| 901820 | Ultraviolet/infra-red ray apparatus | Phototherapy, physiotherapy devices |
| 901831 | Syringes with or without needles | Hypodermic syringes, insulin pens |
| 901832 | Tubular metal needles, sutures | Surgical needles, suture materials |
| 901839 | Other needles, catheters, cannulae | IV catheters, Foley catheters, blood sets |
| 901841 | Dental drill engines | Dental handpieces |
| 901849 | Other dental instruments | Dental tools, endodontic instruments |
| 901850 | Ophthalmic instruments | IOLs, surgical tools, diagnostics |
| 901890 | Other instruments and appliances | Surgical instruments, endoscopes |
Classification note: HS 9018 covers instruments and appliances used in medical, surgical, dental, or veterinary sciences. Active implantable devices (pacemakers) fall under 9021. Orthopaedic implants (plates, screws, prostheses) fall under 9021 as well. Ensure correct classification to avoid customs delays and incorrect duty assessment. Use the HS Code Finder for precise classification.
Quality Standards and Certifications
Regulatory Framework — Device Classification
Medical devices are classified by risk level, and the classification determines the regulatory pathway:
| Class | Risk Level | Examples | USA Pathway | EU Pathway |
|---|---|---|---|---|
| Class I | Low risk | Bandages, tongue depressors, hand instruments | General controls (510(k) exempt for most) | Self-declaration, technical file |
| Class II | Moderate risk | Syringes, catheters, ECG, surgical gloves | 510(k) premarket notification | Notified Body conformity assessment |
| Class III | High risk | Heart valves, pacemakers, implants | PMA (Premarket Approval) | Notified Body clinical evaluation |
US FDA Requirements
The FDA pathway is the most rigorous and most commercially valuable:
Facility Registration: All facilities manufacturing medical devices for the US market must be registered with FDA annually. Registration is done through the FDA Unified Registration and Listing System (FURLS).
510(k) Premarket Notification: Required for Class II devices. You must demonstrate that your device is "substantially equivalent" to a legally marketed predicate device. The submission includes device description, intended use, performance testing, biocompatibility data (if applicable), and labelling. Review timeline: 90 days (statutory), but actual clearance typically takes 6-12 months.
PMA (Premarket Approval): Required for Class III devices. The most stringent pathway — requires clinical trial data demonstrating safety and effectiveness. Timeline: 12-24 months. Costs: $300,000-$1 million+ for the clinical and regulatory process.
Quality System Regulation (QSR) / 21 CFR 820: FDA's GMP requirements for medical devices. Covers design controls, document controls, purchasing controls, production and process controls, corrective and preventive action (CAPA), and complaint handling. FDA inspects manufacturing facilities for QSR compliance.
UDI (Unique Device Identification): All medical devices marketed in the USA must have a UDI — a unique numeric/alphanumeric code on device labels and packages. UDI data is submitted to the FDA's Global Unique Device Identification Database (GUDID).
EU MDR 2017/745 Requirements
The European Union Medical Device Regulation (EU MDR), which replaced the earlier Medical Device Directive (MDD), is now fully in force:
CE Marking: Mandatory for all medical devices sold in the EU. Requires conformity assessment by a Notified Body (for Class II and above).
Technical Documentation: Comprehensive technical file including design documentation, risk management (ISO 14971), clinical evaluation, biocompatibility data, performance testing, and labelling.
Authorised Representative: Non-EU manufacturers must appoint an EU-based Authorised Representative.
Post-Market Surveillance (PMS): Systematic monitoring of device performance after market launch, including vigilance reporting, periodic safety update reports (PSUR), and post-market clinical follow-up (PMCF).
EUDAMED Database: Registration of economic operators, devices, certificates, clinical investigations, and vigilance data in the European Database on Medical Devices.
ISO 13485 — The Foundation
ISO 13485:2016 (Quality Management Systems for Medical Devices) is the foundational quality standard for all medical device exports. It is:
- Required by the EU MDR for CE marking
- Accepted by FDA as evidence of quality system compliance (though FDA inspects against 21 CFR 820)
- Required by most national regulatory authorities worldwide
- The entry ticket to the medical device export business
Indian Regulatory Requirements — CDSCO
The Central Drugs Standard Control Organisation (CDSCO) regulates medical devices in India under the Medical Devices Rules, 2017:
- Manufacturing licence from CDSCO required for all medical devices
- Good Manufacturing Practice (Schedule 5) compliance
- Clinical investigation approval (for Class C and D devices)
- Export NOC from CDSCO for certain controlled devices
Sterilisation Validation
For sterile medical devices (syringes, catheters, sutures, surgical instruments), sterilisation validation is a critical quality requirement:
| Method | Application | Validation Standard |
|---|---|---|
| Ethylene Oxide (EtO) | Most plastic disposables, catheters, sutures | ISO 11135 |
| Gamma Irradiation | Syringes, gloves, drapes, kits | ISO 11137 |
| Steam (Autoclave) | Reusable surgical instruments, textiles | ISO 17665 |
| Electron Beam (E-Beam) | Thin-wall disposables, specialty items | ISO 11137 |
Sterility Assurance Level (SAL) must be 10^-6 (one in a million chance of a non-sterile unit). Sterilisation validation requires biological indicator testing, dose mapping/dose audits, and ongoing routine monitoring.
Biocompatibility Testing
For devices that contact the body, biocompatibility testing per ISO 10993 is mandatory:
- ISO 10993-1: Evaluation and testing framework — determines which tests are needed based on contact type and duration
- ISO 10993-5: Cytotoxicity testing (in vitro)
- ISO 10993-10: Irritation and skin sensitisation
- ISO 10993-11: Systemic toxicity
- ISO 10993-3/6: Genotoxicity and implantation tests (for long-term implants)
Testing must be performed at GLP (Good Laboratory Practice)-compliant laboratories. Major labs: NABL-accredited labs in India, Nelson Labs (USA), WuXi (China), Toxikon (USA).
Packaging and Labelling
Sterile Device Packaging
- Primary packaging: Tyvek/film pouches or blister packs with validated seal integrity
- Peel testing: Seal strength testing per ASTM F88 or EN 868
- Sterile barrier system: Must maintain sterility throughout the shelf life (typically 3-5 years)
- Accelerated ageing studies: Per ASTM F1980 to validate shelf life claims
Labelling Requirements
USA (FDA):
- Device name, model, catalogue number
- Manufacturer name and address
- UDI (Unique Device Identification) in both human-readable and machine-readable (barcode/RFID) format
- "Rx only" or "OTC" designation
- Single-use symbol (if applicable)
- Sterilisation method symbol (EtO, gamma, steam)
- Lot number, manufacturing date, expiry date
- 510(k) or PMA number
- Directions for use (IFU)
EU (MDR):
- UDI-DI (Device Identifier) and UDI-PI (Production Identifier)
- CE mark with Notified Body number
- Manufacturer name and address
- EU Authorised Representative name and address
- Symbols per ISO 15223-1 (internationally recognised medical device symbols)
- Languages: must include the official language(s) of the destination country
Transport Packaging
- Temperature indicators for temperature-sensitive devices
- Shock indicators for sensitive instruments
- Corrugated outer cartons with appropriate handling symbols
- Palletised for container loading, shrink-wrapped
Pricing Strategy
| Product Type | FOB Price Range | Key Factor |
|---|---|---|
| Disposable syringes (3ml, box of 100) | $2-$5 | Volume, sterilisation method |
| IV cannulae (box of 100) | $8-$20 | Gauge, material, safety feature |
| Foley catheters (box of 10) | $5-$15 | Material (latex vs. silicone), size |
| Surgical sutures (dozen) | $8-$30 | Material, needle type, USP size |
| Surgical hand instruments (set) | $50-$500 | Speciality, material (SS vs. titanium) |
| ECG machine (12-lead) | $400-$2,000 | Features, brand, certification |
| Patient monitor (multi-parameter) | $500-$3,000 | Parameters, display, connectivity |
| Laparoscopic instruments (set) | $200-$2,000 | Reusable vs. disposable, complexity |
| IOL (intraocular lens, per unit) | $5-$50 | Material (PMMA vs. hydrophobic acrylic) |
India's pricing advantage is substantial — typically 40-60% below US and European manufacturers for comparable quality devices. For disposable devices (syringes, needles, catheters), the cost advantage is even greater due to lower labour and raw material costs. Indian manufacturers compete with Chinese and Malaysian producers in the disposable segment, and with European and US manufacturers in the higher-value instrument and diagnostic categories.
Margins range from 15-25% for commodity disposables (syringes, needles) to 30-50% for speciality instruments and diagnostic equipment.
Logistics
Shipping
- Air freight: Preferred for high-value devices (diagnostics, implants), temperature-sensitive products, and urgent orders. Mumbai, Delhi, Bangalore, and Hyderabad airports have pharma/medical cargo handling zones.
- Ocean freight: For bulk disposables (syringes, needles, gloves, catheters) shipped in container quantities. 20-ft container holds approximately 20-24 tonnes.
- Temperature-controlled: For temperature-sensitive devices and biological indicators — use GDP-certified logistics providers.
Manufacturing Clusters and Ports
- Faridabad/Ballabhgarh (Haryana) — Disposables hub → ICD Tughlakabad → JNPT
- Pune (Maharashtra) — Instruments and implants → JNPT (3-4 hours)
- Bangalore (Karnataka) — Diagnostics and electronics → Chennai Port or Bangalore ICD
- Chennai (Tamil Nadu) — Sutures and disposables → Chennai Port
- Ahmedabad/Rajkot (Gujarat) — Surgical instruments → Mundra Port
Transit Times
- JNPT to USA East Coast: 28-35 days
- JNPT to Netherlands/Germany: 22-28 days via Suez
- Chennai to Singapore: 8-12 days
- JNPT to Brazil: 25-30 days
- JNPT to UAE: 5-8 days
- JNPT to Turkey: 14-18 days
Freight Costs (Indicative)
- 20-ft container to USA: $2,500-$4,500
- 20-ft container to Europe: $1,800-$3,500
- Air freight (diagnostics/instruments): $4-$10 per kg
- Temperature-controlled air: $8-$15 per kg
Documentation
- Commercial Invoice (with device classification, catalogue numbers, HS codes)
- Packing List (lot/batch-wise, with sterilisation details)
- Bill of Lading / Airway Bill
- Certificate of Origin (from Chamber of Commerce or FIEO)
- Free Sale Certificate (from CDSCO — confirms device is freely sold in India)
- ISO 13485 Certificate (from accredited certification body)
- CE Certificate (for EU-bound shipments, from Notified Body)
- US FDA 510(k) Clearance Letter (for USA-bound shipments)
- Certificate of Analysis / Test Reports (batch-specific)
- Sterilisation Certificate (with SAL confirmation, batch dosimetry records for gamma)
- Biocompatibility Test Reports (as applicable)
- Declaration of Conformity (manufacturer's declaration of regulatory compliance)
- CDSCO Manufacturing Licence and Export NOC
- Shipping Bill (filed via ICEGATE)
- Insurance Certificate
Government Incentives
PLI Scheme for Medical Devices: Production-Linked Incentive scheme provides 5% of incremental sales for specified medical device categories, including:
- Cancer care / radiotherapy devices
- Radiology and imaging devices (CT, MRI)
- Anaesthesia and cardio-respiratory devices
- Implants (orthopaedic, cardiac)
- Renal care devices
Medical Device Parks: The government is developing four medical device manufacturing parks (Andhra Pradesh, Himachal Pradesh, Madhya Pradesh, Tamil Nadu) with shared infrastructure including sterilisation facilities, testing labs, and warehousing.
RoDTEP: Remission of Duties and Taxes at 0.5-2% of FOB value for medical device exports.
EPCG Scheme: Duty-free import of capital goods (clean room equipment, sterilisation plants, testing instruments) against export obligations.
AIDMMA Support: Association of Indian Medical Device Manufacturers Association provides market intelligence, regulatory guidance, and trade fair participation support.
Pharmexcil: Pharmaceutical Export Promotion Council also covers medical devices — RCMC from Pharmexcil is required for claiming export incentives.
Common Mistakes to Avoid
Treating regulatory approval as a one-time event. FDA 510(k) clearance and CE marking are the beginning, not the end. Post-market surveillance, adverse event reporting, complaint handling, and periodic regulatory updates are ongoing obligations. A single unreported adverse event can trigger an FDA Warning Letter or CE certificate suspension.
Inadequate design controls. FDA's 21 CFR 820 and EU MDR require formal design controls — design input, output, verification, validation, and design transfer. Many Indian MSME manufacturers skip the design control process and go straight to production. This works until an FDA inspector asks for your design history file. Invest in formal design and development procedures from the start.
Failing sterilisation validation. An improperly validated sterilisation process can release non-sterile "sterile" devices into the market — a patient safety catastrophe. Sterilisation validation must follow the applicable ISO standard (11135 for EtO, 11137 for radiation, 17665 for steam), with documented biological indicator testing, dose mapping, and routine monitoring. Never shortcut this process.
Not understanding device classification. Misclassifying your device — for example, treating a Class II device as Class I — leads to marketing without the required premarket notification. This is a regulatory violation that can result in product seizure, injunction, and import detention in the USA, or market withdrawal in the EU. Consult CDSCO, FDA, and EU classification rules carefully.
Ignoring UDI requirements. The Unique Device Identification system is now mandatory in the USA, EU, and an increasing number of countries. Each device must carry a UDI label, and device data must be submitted to the relevant database (GUDID in the USA, EUDAMED in the EU). Non-compliance results in market access denial.
Key Takeaways
- India exported $1,355.74 million in surgical instruments (HS 9018) in 2024-25 — nearly doubling from $700M in 2022-23
- Syringes/needles/catheters ($512.7M) and other instruments ($478.6M) are the two largest categories
- USA ($280.8M), Netherlands ($88.0M), and Germany ($80.6M) are the top three markets
- ISO 13485 certification is the absolute foundation — without it, no export market is accessible
- US FDA 510(k) clearance is required for Class II devices and is the most commercially valuable regulatory approval
- EU MDR 2017/745 requires CE marking, Notified Body assessment, and EU Authorised Representative
- Sterilisation validation (ISO 11135/11137/17665) and biocompatibility testing (ISO 10993) are non-negotiable for sterile devices
- India's 40-60% cost advantage over Western manufacturers makes it highly competitive
- PLI scheme and medical device parks are accelerating India's manufacturing capability
Next Steps
- Classify your device accurately using the HS Code Finder — HS 9018 covers surgical instruments, but related devices may fall under 9019, 9021, or 9022
- Obtain ISO 13485 certification — this is the non-negotiable first step for any medical device export
- Determine your regulatory pathway for each target market — 510(k) for USA, CE marking under MDR for EU, CDSCO registration for India
- Invest in sterilisation validation and biocompatibility testing — partner with GLP-certified labs and ISO 17025-accredited testing facilities
- Research target markets with the Market Finder — identify which device categories are in demand in each country
- Check tariff rates using the Duty Calculator — medical devices face 0-5% duty in most markets, with many enjoying zero-duty under ITA agreements
- Register with Pharmexcil for RCMC and access to export promotion support
- Attend trade fairs — MEDICA (Dusseldorf), Arab Health (Dubai), FIME (Miami), Medical Fair India (Delhi)
The global medical device market is projected to reach $800 billion by 2030. India, with its combination of engineering talent, cost competitiveness, and expanding regulatory infrastructure, is positioned to become one of the world's top five medical device exporters within this decade. The MSME manufacturers who invest in quality systems, regulatory compliance, and clinical evidence today will be the ones supplying hospitals worldwide tomorrow.
Ready to start exporting?
Get a detailed market report with country-by-country analysis, pricing insights, and buyer contacts.
Get Market ReportXIMPEX Export Advisory
Need help implementing the strategies discussed in this guide? Our team of export specialists can provide personalized guidance for your business.