How to Export Ayurvedic and Herbal Products from India — Complete Guide
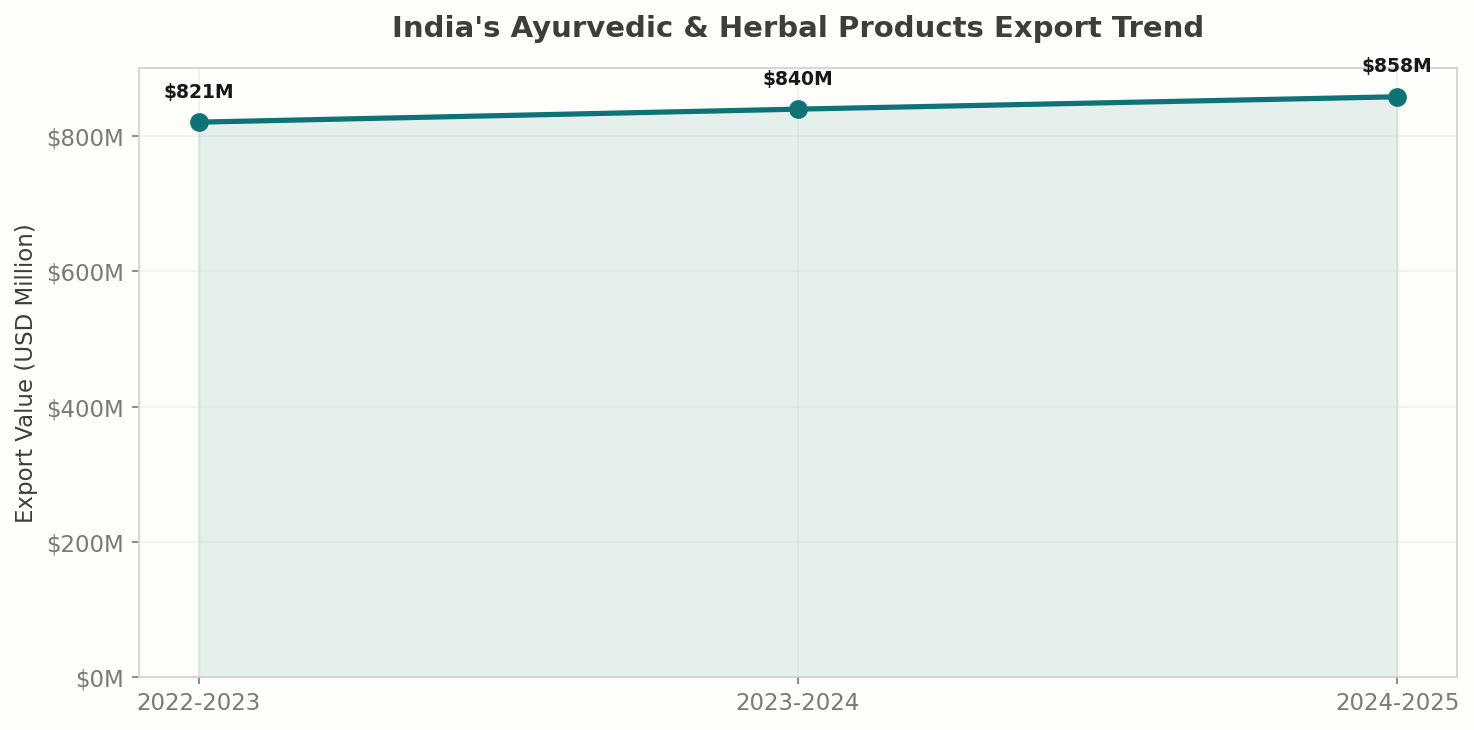
India is the global epicentre of Ayurveda and traditional herbal medicine. In 2024-25, India's exports of ayurvedic and herbal products reached $858 million, up from $820 million in 2022-23 — a steady 4.6% increase driven by rising global demand for natural health and wellness products. India supplies medicinal herbs, botanical extracts, ayurvedic formulations, and herbal supplements to over 100 countries, with the United States, Germany, and Italy as the leading buyers.
For MSME exporters — whether you are a manufacturer of ayurvedic formulations in Kerala, a herbal extract processor in Madhya Pradesh, or a trader sourcing raw botanicals from Uttarakhand — this sector offers significant margins and growing international acceptance. However, the regulatory landscape is complex. An ayurvedic product classified as a "dietary supplement" in the USA faces entirely different rules than the same product classified as a "traditional herbal medicinal product" in the EU or a "drug" under Indian law. This guide covers the regulatory pathways, quality requirements, market access strategies, and practical logistics for exporting ayurvedic and herbal products from India.
India's Ayurvedic and Herbal Product Export Landscape
India exported $858 million in ayurvedic and herbal products in 2024-25, continuing a growth trajectory from $820 million in 2022-23. The global herbal medicine market is valued at over $300 billion and is growing at 8-10% annually, driven by consumer preference for natural remedies, clean-label products, and preventive healthcare.
Indian ayurvedic and herbal exports are classified primarily under two HS headings:
| HS Code | Category | 2024-25 Exports (USD Million) | Share |
|---|---|---|---|
| 1211 | Plants, seeds, and herbs used in pharmacy or perfumery | $533 | 62.1% |
| 3003 | Medicaments consisting of mixed herbs (not in dosage form) | $325 | 37.9% |
Raw and semi-processed herbs and botanicals (HS 1211) dominate at $533 million, reflecting strong global demand for Indian medicinal plants like ashwagandha, turmeric, tulsi, neem, brahmi, and shatavari. Herbal medicaments (HS 3003) at $325 million represent formulated ayurvedic products — churnas, kwaths, arishtas, and herbal extracts in bulk form.
Where Indian Ayurvedic and Herbal Products Are in Demand
Indian ayurvedic and herbal products are exported to over 100 countries, with particularly strong demand in three clusters. Western markets — led by the USA and Germany — drive the largest share of exports, fuelled by the booming dietary supplements and wellness industry where ingredients like ashwagandha, turmeric/curcumin, and brahmi have become mainstream. Europe, particularly Germany, Italy, and Belgium, is a major destination for botanical raw materials and standardised herbal extracts used in pharmaceutical and nutraceutical manufacturing. The Middle East and South Asian diaspora markets provide consistent demand for traditional ayurvedic formulations — churnas, tailams, and classical medicines.
Want the full country-by-country breakdown? See exact export values, growth rates, tariff rates, and market attractiveness scores for every destination in our detailed data pages. View Ayurvedic and Herbal Products export data by country →
Product Categories
Raw Herbs and Botanicals (HS 1211)
This is the largest export category. India exports dried whole herbs, roots, seeds, leaves, bark, and flowers used in pharmaceutical, nutraceutical, and cosmetic manufacturing worldwide. Key exports include:
- Ashwagandha (Withania somnifera) — roots and root extracts
- Senna (Cassia angustifolia) — leaves and pods, used as a natural laxative
- Psyllium husk (Plantago ovata) — major export from Gujarat
- Turmeric (Curcuma longa) — both whole and as curcumin extract
- Brahmi (Bacopa monnieri) — for cognitive health supplements
- Neem (Azadirachta indica) — leaves, bark, oil
- Tulsi (Ocimum sanctum) — dried leaves and extracts
Ayurvedic Formulations (HS 3003)
Classical ayurvedic formulations and proprietary herbal medicines in bulk form:
- Churnas — herbal powders (Triphala, Ashwagandha, Brahmi)
- Kwaths/Kashayams — herbal decoctions
- Arishtas and Asavas — fermented herbal liquids
- Ghritams — medicated ghee preparations
- Tailams — medicated oils (Mahanarayan, Bhringraj)
- Proprietary formulations — branded herbal supplements and combinations
HS Code Classification
Correct classification determines the regulatory pathway in the destination country. The distinction between a "food supplement" and a "medicine" often hinges on the HS code and product claims:
| HS Code | Description | Typical Products |
|---|---|---|
| 1211 | Plants and parts of plants used in pharmacy, perfumery, or insecticidal purposes | Dried herbs, roots, seeds, leaves, bark |
| 121190 | Other plants and parts — mixed herbs | Herb blends, mixed botanicals |
| 3003 | Medicaments of two or more constituents mixed together, not in dosage form | Bulk herbal formulations, churnas, extracts |
| 300390 | Other medicaments of mixed constituents | Ayurvedic classical formulations in bulk |
| 3004 | Medicaments in measured doses or for retail sale | Packaged ayurvedic tablets, capsules, syrups |
| 1302 | Vegetable saps and extracts | Standardised herbal extracts (curcumin, bacosides) |
| 2106 | Food preparations not elsewhere specified | Herbal food supplements, health drinks |
Critical classification issue: If your product makes therapeutic claims (e.g., "cures arthritis"), it will be classified as a medicine in most countries and require drug registration. If it makes structure/function claims (e.g., "supports joint health"), it may qualify as a dietary supplement with lighter regulation. The HS code must align with the product's regulatory positioning. Use the HS Code Finder for precise classification.
Quality Standards and Certifications
Domestic Regulatory Requirements
AYUSH Ministry Registration: The Ministry of AYUSH governs the manufacture and regulation of ayurvedic, siddha, unani, and homeopathic medicines in India. Any ayurvedic product intended for export must be manufactured in a facility licensed under the Drugs and Cosmetics Act, 1940 (for products classified as drugs) or by a manufacturer with a valid AYUSH manufacturing licence.
Drug vs. Food Classification in India: This is the fundamental regulatory question:
- Ayurvedic drug: Products listed in the Ayurvedic Pharmacopoeia or manufactured as per classical formulations require a manufacturing licence from the State Drug Controller under AYUSH.
- Food/supplement: Products positioned as health supplements, nutraceuticals, or foods for special dietary uses fall under FSSAI regulations (FSSAI Act, 2006 and FSS Regulations on Health Supplements, 2016).
FSSAI Licence: Required for all food-category herbal products. Central FSSAI licence if annual turnover exceeds Rs 12 crore or if you export. Product approval is required for novel ingredients not listed in FSSAI schedules.
International Certifications
GMP/WHO-GMP: Good Manufacturing Practice certification is the baseline for all markets. WHO-GMP certification from the AYUSH Ministry or CDSCO demonstrates international-standard manufacturing. Many importing countries require a GMP certificate as a condition of import.
Organic Certification: Increasingly important for herbal exports. NPOP (National Programme for Organic Production) certification is recognised in the EU, USA (NOP equivalent), and other markets. Certifying agencies include India Organic, APEDA-approved bodies, and international certifiers like Ecocert, USDA NOP, and EU Organic.
Ayush Premium Mark: Launched by the Quality Council of India, this voluntary certification provides quality assurance for ayurvedic products with testing for identity, purity, strength, and safety. It is gaining recognition in export markets as a quality differentiator.
ISO 22000 / FSSC 22000: Food safety management system certification, useful for herbal products classified as foods or supplements.
Market-Specific Regulatory Requirements
USA — FDA DSHEA Compliance: The USA classifies most herbal products as dietary supplements under the Dietary Supplement Health and Education Act (DSHEA, 1994). Key requirements:
- Facility must be registered with FDA
- Products must comply with 21 CFR Part 111 (cGMP for dietary supplements)
- New Dietary Ingredient (NDI) notification required for ingredients not marketed before October 1994
- Supplement Facts label (not Nutrition Facts)
- Structure/function claims only — no disease claims without drug approval
- Adverse event reporting mandatory
EU — Traditional Herbal Medicinal Product Directive (THMPD): The EU has a specific pathway for traditional herbal medicines under Directive 2004/24/EC:
- Product must demonstrate 30 years of traditional use (15 years within the EU)
- Simplified registration — no clinical trial data required
- Bibliographic evidence of safety and efficacy
- GMP compliance mandatory
EU — Novel Food Regulation: Herbal ingredients without a history of significant consumption in the EU before May 1997 are classified as "novel foods" under Regulation (EU) 2015/2283 and require pre-market authorisation. Many traditional Indian herbs (ashwagandha was approved in 2022, but others remain under review) face this barrier. Check the EU Novel Food Catalogue before exporting.
Canada — Natural Health Products Regulations: Health Canada regulates herbal products as Natural Health Products (NHPs). Requirements include:
- Product licence with an NPN (Natural Product Number)
- Evidence of safety and efficacy
- GMP compliance (validated by a site licence)
- Proper labelling in English and French
Critical Testing Parameters
Heavy Metal Testing: This is the single biggest quality risk for Indian herbal products. International limits:
- Lead: <3 ppm (USA), <3 mg/kg (EU)
- Mercury: <1 ppm (USA), <0.1 mg/kg (EU)
- Arsenic: <10 ppm (USA), <5 mg/kg (EU Pharmacopoeia)
- Cadmium: <0.3 mg/kg (EU)
Pesticide Residues: EU has the strictest MRLs (Maximum Residue Limits). Many Indian herbs fail EU pesticide testing due to agricultural practices. Ethylene oxide has zero tolerance in the EU — a common cause of Indian herbal product rejections at EU borders.
Microbial Contamination: Total aerobic count, yeasts and moulds, E. coli, Salmonella, and Staphylococcus aureus limits apply. Standards vary by pharmacopoeia (USP, EP, IP).
Aflatoxin Levels: EU limit: 2 ppb for aflatoxin B1, 4 ppb total in herbal products intended for direct consumption.
Packaging and Labelling
Packaging Requirements
- Raw herbs (bulk): Multi-wall kraft paper bags with food-grade HDPE inner liners (25-50 kg). Vacuum-sealed or nitrogen-flushed for oxidation-sensitive herbs.
- Herbal extracts: HDPE drums (25-50 kg) with double-sealed lids. Food-grade, moisture-proof.
- Finished formulations: HDPE bottles, glass bottles, aluminium blisters (for tablets/capsules), laminated sachets (for powders). Child-resistant closures for USA market.
- Retail packs: Multi-layer laminate pouches (BOPP/foil/PE) for powders and teas. UV-protective packaging for light-sensitive herbs.
Labelling Requirements
USA (FDA):
- Supplement Facts panel (for dietary supplements)
- Identity statement ("Dietary Supplement")
- Net quantity
- Name and address of manufacturer/distributor
- Complete ingredient list with amounts
- "This statement has not been evaluated by the FDA" disclaimer for structure/function claims
- Allergen declarations (if applicable)
EU:
- Product name and botanical name (Latin binomial)
- Net quantity
- Ingredients list with quantitative declaration
- Allergen information
- Best-before date
- Storage conditions
- Name and address of EU-based responsible party
- Batch/lot number
- Country of origin
- Nutritional declaration (for food-category products)
General:
- All text in the destination country's language(s)
- Avoid therapeutic/disease claims unless the product is registered as a medicine
- "Made in India" or "Product of India"
Pricing Strategy
Pricing depends heavily on the product form, standardisation level, and certification status:
| Product Type | FOB Price Range | Key Driver |
|---|---|---|
| Dried whole herbs (bulk) | $2-$15/kg | Species, organic certification |
| Herbal powders (bulk) | $5-$25/kg | Fineness, purity |
| Standardised extracts | $30-$200/kg | Active compound concentration |
| Organic certified herbs | 30-60% premium over conventional | Certification costs, lower yields |
| Finished ayurvedic formulations (bulk) | $10-$50/kg | Formulation complexity, GMP |
| Retail-packed supplements (capsules) | $0.50-$3.00/bottle | Branding, market positioning |
| Ashwagandha extract (5% withanolides) | $50-$120/kg | Standardisation level |
| Curcumin extract (95%) | $80-$180/kg | Curcuminoid content |
Margins for standardised extracts (25-45%) are significantly higher than raw herbs (10-20%). Moving up the value chain from raw botanicals to standardised, certified extracts dramatically improves profitability.
Logistics
Shipping Considerations
Most herbal products ship via ocean freight in standard dry containers. Temperature-sensitive extracts and live cultures may require temperature-controlled containers (15-25 degrees Celsius).
Primary Export Ports
- JNPT/Nhava Sheva (Maharashtra) — Primary port for herbal product exports
- Mundra (Gujarat) — For psyllium, senna, and Rajasthan/Gujarat-sourced herbs
- Kochi (Kerala) — For South Indian ayurvedic products and spice-derived herbs
- Chennai — For South Indian herbal manufacturers
Transit Times
- JNPT to USA East Coast: 28-35 days
- JNPT to EU ports (Hamburg, Rotterdam): 20-26 days via Suez
- Mundra to UK: 18-22 days
- Kochi to UAE: 5-7 days
Freight Costs (Indicative)
- 20-ft container to USA: $2,500-$4,500
- 20-ft container to Europe: $1,800-$3,500
- 20-ft container to UAE: $800-$1,500
- Air freight (samples/urgent): $4-$8 per kg
Container Capacity
- Dried herbs in bags: 14-18 tonnes per 20-ft container
- Herbal extracts in drums: 12-16 tonnes per 20-ft container
Documentation
Complete documentation for an ayurvedic/herbal product export consignment:
- Commercial Invoice (with botanical names, HS codes, batch numbers)
- Packing List
- Bill of Lading / Airway Bill
- Certificate of Origin (from Chamber of Commerce or FIEO)
- Phytosanitary Certificate (from Plant Quarantine Division for raw herbs)
- Certificate of Analysis (CoA) — heavy metals, pesticide residues, microbial, identity
- GMP/WHO-GMP Certificate
- AYUSH Manufacturing Licence (for ayurvedic drug products)
- FSSAI Licence copy (for food-category products)
- Organic Certificate (NPOP/NOP/EU Organic, if applicable)
- Health Certificate (required by some countries)
- Free Sale Certificate (from State Drug Controller or CDSCO)
- CITES permit (for any species listed under CITES — e.g., certain Aloe species, Red Sanders)
- Shipping Bill (filed via ICEGATE)
For the EU, additionally prepare a pesticide residue analysis report, aflatoxin report, and heavy metal analysis from an accredited laboratory.
Government Incentives
AYUSH Ministry Export Promotion: The Ministry of AYUSH actively promotes international acceptance of ayurvedic products through bilateral agreements, participation in international health forums, and the Ayush Premium Mark programme.
APEDA Support: The Agricultural and Processed Food Products Export Development Authority provides financial assistance for quality upgrades, organic certification, and participation in international trade fairs for botanical and herbal products.
RoDTEP (Remission of Duties and Taxes on Exported Products): Refunds embedded taxes at 0.5-3% of FOB value, depending on the HS code.
Transport and Marketing Assistance (TMA): Financial support for freight costs to new and emerging markets, particularly in Africa, Latin America, and Central Asia.
PLI Scheme for Pharmaceuticals: For herbal formulations classified as drugs, the PLI scheme provides incentives of 5-20% on incremental sales of specified products.
National AYUSH Mission: Provides support for cultivation of medicinal plants, establishment of quality testing laboratories, and technology upgradation for AYUSH manufacturing units.
MPEDA and Spices Board: For specific herbal products that overlap with spice categories (turmeric, ginger), additional support is available through these commodity boards.
Common Mistakes to Avoid
Misclassifying your product. The line between a "food supplement" and a "medicine" varies by country. Selling an ayurvedic medicine as a dietary supplement in the USA without proper classification can trigger FDA enforcement action. Conversely, unnecessarily registering a simple herbal tea as a medicine adds years of delay and hundreds of thousands of dollars in regulatory costs. Get professional regulatory advice for each target market.
Ignoring heavy metal contamination. Indian soil and water can contain naturally elevated levels of lead, arsenic, and mercury. Ayurvedic formulations using bhasmas (metal-based preparations) have been specifically flagged by the FDA and EU authorities. Every batch must be tested. A single contaminated shipment can destroy your brand reputation and result in an import alert.
Not checking Novel Food status in the EU. Many traditional Indian herbs are classified as "novel foods" in the EU because they lack a documented history of consumption in Europe before 1997. Exporting ashwagandha was restricted until its Novel Food approval in 2022. Check the EU Novel Food Catalogue for every ingredient before shipping to Europe. Unauthorised novel food ingredients will be seized at the border.
Failing pesticide residue testing. Herbs sourced from conventional agriculture often fail EU pesticide MRLs. Ethylene oxide, used for sterilisation, has zero tolerance in the EU. Invest in organic sourcing or implement rigorous pesticide-free cultivation protocols. Test before you ship — a test costs Rs 10,000-25,000; a rejected container costs Rs 10-20 lakh.
Making therapeutic claims on labels. Claiming "cures diabetes" or "treats cancer" on a product sold as a food supplement is illegal in virtually every market. Even in India, such claims on food products violate FSSAI regulations. Restrict claims to structure/function language ("supports healthy blood sugar levels") and ensure they comply with destination country advertising laws.
Key Takeaways
- India exported $858 million in ayurvedic and herbal products in 2024-25, up 4.6% from 2022-23
- Raw herbs/botanicals (HS 1211) at $533M and herbal medicaments (HS 3003) at $325M are the two primary categories
- USA ($217.9M), Germany ($116.4M), and Italy ($43.9M) are the top three markets
- Regulatory classification (drug vs. supplement vs. food) determines the entire export pathway — get this right first
- Heavy metal testing, pesticide residue analysis, and microbial testing are non-negotiable for every batch
- EU Novel Food regulation is a major barrier — verify ingredient status before targeting European markets
- WHO-GMP certification from AYUSH Ministry is the baseline for credible international market access
- Organic certification commands 30-60% price premiums and opens doors in quality-conscious markets
- Standardised herbal extracts offer 25-45% margins vs. 10-20% for raw herbs
Next Steps
- Identify your exact HS code using the HS Code Finder — classification determines your regulatory pathway in every market
- Determine your product's regulatory status in your target market — food supplement, traditional herbal medicine, or drug
- Obtain WHO-GMP certification for your manufacturing facility from the AYUSH Ministry or CDSCO
- Invest in laboratory testing — partner with NABL-accredited labs (SGS, Eurofins, Intertek) for heavy metals, pesticides, microbial, and identity testing
- Research target market requirements with the Market Finder — demand patterns differ significantly between the USA (supplements), EU (traditional herbal medicines), and Middle East (ayurvedic medicines)
- Check duty rates and trade agreements using the Duty Calculator — herbal products face 0-12% duty depending on classification and market
- Consider organic certification (NPOP) — the investment pays for itself through price premiums and market access
The global shift toward natural, plant-based health products is structural and accelerating. With its 5,000-year-old ayurvedic tradition, unmatched biodiversity of medicinal plants, and growing manufacturing capability, India is uniquely positioned to lead this $300 billion market. The exporters who invest in quality certification, regulatory compliance, and product standardisation will capture the lion's share of this opportunity.
Ready to start exporting?
Get a detailed market report with country-by-country analysis, pricing insights, and buyer contacts.
Get Market ReportXIMPEX Export Advisory
Need help implementing the strategies discussed in this guide? Our team of export specialists can provide personalized guidance for your business.